 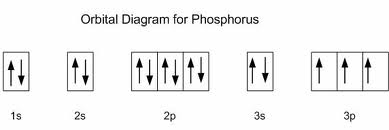
The ground state electron configuration of ground state gaseous neutral cadmium is Kr. Mårtensson, "Core-Level Binding Energies in Metals," J. Cadmium atoms have 48 electrons and the shell structure is 2.8.18.18.2. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). So, we have Calcium Electron Configuration is: 1s² 2s² 2p 3s² 3p 4s². The electron configuration for antimony is 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 10. The 6 electrons will go to the 2p orbital, and the next 2 electrons will place with the 3s orbital, and now we have only 8 electrons in which 6 electrons will go with the 3p orbital, and the last 2 electrons will be with the 4s orbital. The data are adapted from references 1-3. In quantum chemistry and atomic physics, the electron configuration of an. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. All values of electron binding energies are given in eV. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cadmium. These effective nuclear charges, Z eff, are adapted from the following references: 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
